Use of FRI in deposition of inhaled lip. cyclosporin in BOS
Use Of Functional Respiratory Imaging (FRI) To Explore Drug Delivery And Lung Deposition Of Inhaled Liposomal Cyclosporine A (L-CsA-i) In Patients After Lung Transplantation (LTx)
FRI-based biomarkers have been investigated as early indicators of bronchiolitis obliterans syndrome (BOS) in lung transplant patients. BOS is a progressive obstructive airway disease characterized by inflammation and fibrosis that results in respiratory failure and death. Currently there are no approved therapies for BOS.
The aim of this study was to characterize the airway deposition of Liposomal cyclosporine-A for inhalation (L-CsA-i) in lung transplant recipients with BOS using FRI.
Results:
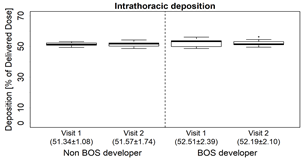
- Intrathoracic deposition was similar at both time points in both BOS and non-BOS developers, (Fig. 2A)
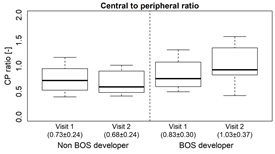
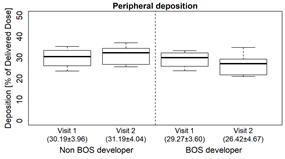
- Of the deposited dose, patients who did not develop BOS had a slight increase in deposition to the peripheral airways, while the BOS group has a slight decrease in deposition to the peripheral airways (Fig. 2B) A similar change was also seen in the central to peripheral ratio (fig.2C).
Conclusions:
- FRI has potential to become a useful biomarker for BOS patients and might be useful to characterize aerosolized L-CsA-i deposition
- FRI shows that L-CsA-i, a drug under development for the treatment of BOS, has >50% deposition when delivered via an optimized Investigational eFlow® Technology nebulizer system to target airways
- FRI also shows that L-CsA-i can reach affected airways at similar concentrations as unaffected airways

Categorised in: Miscellaneous / August 7, 2020 10:44 am /
Tags: FRI, Poster