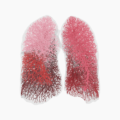
Fluidda’s Broncholab issued Authorization with Conditions by Health Canada for Use in Relation to COVID-19
May 28, 2021 11:19 am Leave your thoughtsIn March 2020 Fluidda’s medical device called Broncholab was introduced in the USA. It is a diagnostic support tool to detect and to to improve therapy of lung diseases based on FRI technology. Broncholab will initially... View Article